KANSAS CITY, Mo. — This week, the state of Michigan marked a grim milestone--a record high for the number of children hospitalized with COVID-19.
At this point there are also more than 300 ongoing outbreaks tied to schools in the state.
Health officials say the highly contagious B.1.1.7 variant is spreading quickly.
It's something Smithville, Missouri, mom Amanda Maynes is watching closely, especially since her kids recently returned to in-person learning.
The good news is that her eldest, 17-year-old Rhiannon, already received the vaccine. Delainy, her middle daughter, is about to celebrate her sweet 16 with a dose.
"She might have her vaccine before she gets her driver's license test," Maynes said while laughing.
However, Avery, her eighth grader, hasn't had the same opportunity.
"My 14-year-old has no way to be vaccinated yet," Maynes said.
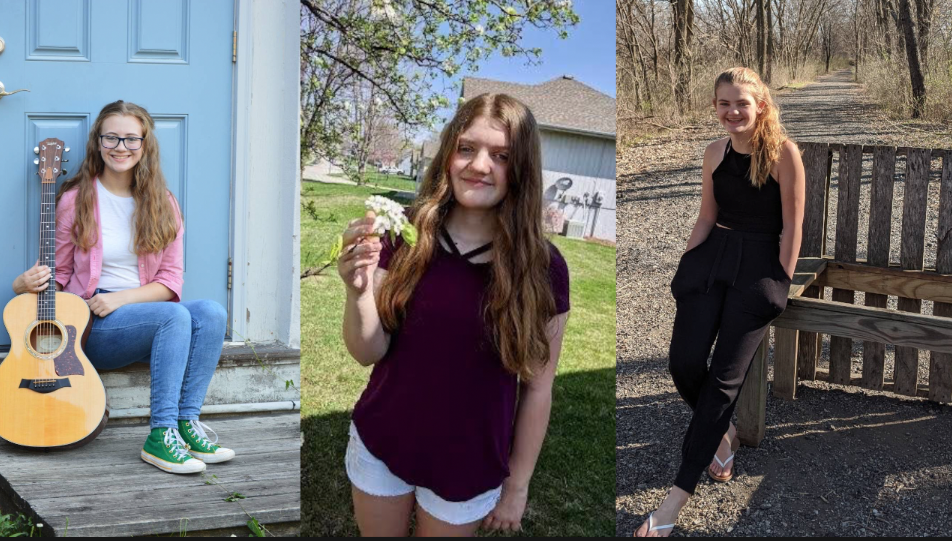
The FDA's current emergency use authorization for the Moderna vaccine covers only adults, and the Pfizer vaccine is cleared for people ages 16 and up.
However, that could soon change. Pfizer is asking the agency to amend its EUA to include kids ages 12-15.
Pfizer recently announced a trial showed the vaccine was 100% effective in preventing infection in that age group.
According to experts interviewed by NBC, the FDA's review process will probably take several weeks.
"While the FDA cannot predict how long our evaluation of the data and information will take, we will review the request as expeditiously as possible using our thorough and science-based approach," FDA Commissioner Dr. Janet Woodcock tweeted last Friday.
As with all FDA-authorized #COVID19 vaccines, we are committed to transparency with this EUA review process.
— Dr. Janet Woodcock (@DrWoodcockFDA) April 9, 2021
The initial EUA for the Pfizer vaccine was recommended at a Vaccines and Related Biological Products Advisory Committee meeting in December 2020.
"Based on an initial evaluation of the information submitted, at this time the agency does not plan to hold a meeting of the VRBPAC on this request to amend the EUA," FDA Press Officer Abby Capobianco wrote in an email to 41 Action News.
The agency again said it could not predict how long the review process will take.
Maynes is watching closely and hoping the green light comes soon.
"I will be elated," she said. "I will be right back on this computer filling out the forms and getting Avery signed up."
It's a decision Maynes said her daughter researched and made on her own.
"She realizes that this is better not just for her, but also for everybody around her, too," Maynes said.





